Some of the defining features of batteries are Nominal Voltage, Specific Energy, State-of-charge (SOC). This article talks about the battery capacity or more precisely the C-Rating.
Definition
The C rate of A battery cell represents the level of constant current charge or discharge that the cell can sustain for one hour of time.
— OR —
C rating is defined as a rating of how quickly energy is discharged from a battery.
— OR —
C-rates govern charge and discharge rates of a battery.
— OR —
C in C rate stands for the capacity of a battery usually measured in ampere-hours (Ah) indicating the amount of active material within the battery available for discharge.
Let's Explore
For example, a cell having a capacity of 20-ampere hours should be able to deliver 20 amperes of current for one hour or 2 amperes of current for 10 hours. The C rate may be a way of normalizing electrical current in line with a cell’s nominal capacity. 
The capacity of A battery is often rated at 1C, meaning that a charged battery rated at 1Ah should provide 1A for one hour. The same battery discharging at 0.5C should provide 500mA for 2 hours, and at 2C it delivers 2A for a half-hour. Losses at fast discharges reduce the discharge time, and these losses also affect charge times.
A C-rate of 1C is additionally referred to as a one-hour discharge; 0.5C or C/2 is a two-hour discharge and 0.2C, or C/5 is a 5-hour discharge. Some high-performance batteries are often charged and discharged above 1C with moderate stress. Table 1 illustrates the typical times at various C-rates.
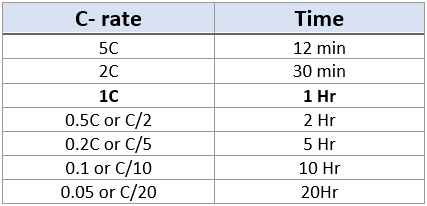
Table 1: C-rate and time of charge or discharge 1Ah Battery cell. [1]
When discharging a battery with a battery analyser capable of applying different C rates, a higher C rate will produce a lower capacity reading and vice versa. By discharging the 1Ah battery at the faster 2C-rate, or 2A, the battery should ideally deliver the full capacity in half-hour. The sum should be the same since the identical amount of energy is dispensed over a shorter time. In reality, internal losses turn some part of the energy into heat and lower the resulting capacity to about 95 per cent or less. Discharging a similar battery at 0.5C, or 500mA over 2 hours, will likely increase the capacity to above 100%.
C-Rating Calculation
Let us consider A 20C battery rated at 2000 mAh. To calculate what this battery can do. Convert milliamps to amps:
2000mAh = 2000 * 10–3 Ah = 2 Ah
Now, Calculate the amps times the C-rating to get the total output current:
Total Output Current = 2 Ah * 20C = 40 A
This means that we can discharge 40 amps continuously without damaging the battery.
C-Rate Commercially
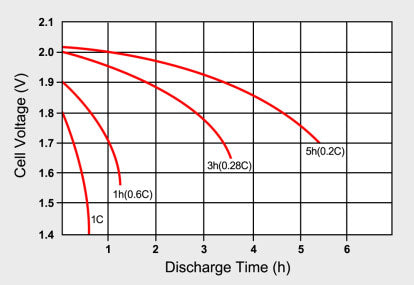
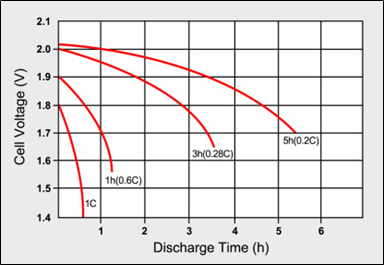
To obtain a reasonably good capacity reading, manufacturers commonly rate alkaline and lead-acid batteries at an awfully low 0.05C or a 20-hour discharge. Even at this slow discharge rate, The lead-acid batteries seldom attain a 100 per cent capacity as the batteries are overrated. Manufacturers provide capacity offsets to regulate for the discrepancies if discharged at a higher C rate than specified. Figure 2 illustrates the discharge times of a lead-acid battery at various loads expressed in C-rate. Smaller batteries are rated at a 1C discharge rate. Due to sluggish behaviour, lead-acid is rated at 0.2C (5h) and 0.05C (20h).
C- Rate of batteries used for drones
Drones are unquestionably one of the greatest innovations of our time. These flying machines are used in almost all sectors today. Call it for surveillance, delivery at remote locations or even photography and recreational doings they are used everywhere. For better experiences, we require batteries with adequate capacity and good C-rating. It is instinctively recommended to use the battery of highest available C-rating.

The markets are flooded with lots of good C-rating batteries. A battery that has a higher C rating delivers more energy, and that means higher performance. The big difference between batteries with different C ratings is the fact that the higher-rated batteries suffer less from voltage drops under load.
Let us take the example which we considered earlier of 20C battery rated at 2000mAh. Concerning the calculation of output current, we know that this battery can happily discharge 40 A, without demanding a charge for say 2 hrs.
However, now if we try to fly the drone with the same battery for much longer say 4 hrs, then the battery cannot give expected output. Similarly, if we increase the payload of the drone or install one more camera to it, then the discharge from the battery will be much more, leading to zero charges in the battery.
There are battery packs in the markets rated at 30C, 45C, and even 70C that will deliver (in our 2000mAh example) 60, 90, and 140 amps. Which not only gives you the chance to enhance the features of your drones but also saves your time as these charges fast.
Sources:
- https://batteryuniversity.com/learn/article/what_is_the_c_rate
- quadquestions.com
- google.com
- energsoft.com
- researchgate.net
- cleanenergywiki.org
- https://www.rotordronepro.com/
- https://images.app.goo.gl/48dcwue3ypZJh3Hu8
- https://images.app.goo.gl/6gbkZZVvZZKXmAGB7
- https://images.app.goo.gl/4LxpGpEEWbMqyZwdA